SAS® Savvy e-Guides, Books, Self-Study Classes, and One-on-One Mentoring

Interested in getting more SAS® resources for an in-depth SAS® programming knowledge and understanding? Consider my popular SAS® e-Guides, SAS® Books, public SAS® training on-demand classes, Pharmaceutical Industry on-demand classes, or my One-on-One mentoring program. SAS® Savvy Premium/University Student members have automatic access to SAS® e-Guides and SAS® training on various topics introduced on a monthly basis.
Not a SAS® Savvy Premium/University Student member? There is still time to join now for all of the benefits.
Partial list of our satisfied clients: Amgen, Pfizer, Edwards LifeSciences, Ionis, BioMarin, Omeros, BioBridges, Synteract, Averon, MarketShare, Connetics, Kaiser Permanente, Judicial Council of California, and Quintiles.
- One-on-One_Mentoring
- SAS_Programming_Training_Classes
- SAS_Books
- Download_Popular_SAS_e-Guides
- Pharmaceutical_Industry_On-Demand_Classes
Looking to make a career transition from the health care or research related field into the pharmaceutical industy? E-mail us for more information about my One-on-One Mentoring program.

SAS Programming Training Classes (Top)
"Hello Dr. Gupta, I attended your webinar today on Proc Tabulate along with 15 of my colleagues here at the U of Utah. We were all very impressed and found many useful pointers. Thanks", Molly McFadden, MS, Unversity of Utah, ASA Member, Feb. 2013
"Your course exactly connects with the practical jobs in SAS programming of Pharmaceutical industry. I enjoyed learning it, and it gave me back my confidence for seeking my new career development in future. It's a great help! Thanks to Mr. Gupta for sharing his valuable experience. It's exactly what I want to learn. I hope I could learn some more from you.", Thanks!, Your student, Yuhong Huang
At SAS® conferences, students have paid hundreds of dollars for a four-hour class and thousands for an eight-hour SAS classes. Email us if you want to download these self-study SAS® class course notes and SAS®examples for a member discount price to save over 30%. This includes a several one-on-one web sessions to get you started and answer questions and limited e-mail support. Selected self study classes include e-guides which are valued up to $45 each.
The same three-days live CDISC corporate class worth of content contains practical examples includes all class slides, SDTMs and ADaMs, SAS macros, a total of four up to thirty-minute one-on-one web sessions, a unique 40 page e-guide, access to five video recordings of CDISC classes and nine CDISC portals to access industry expert presentations. Students study 1) mapping to SDTM and ADaM steps, 2) compare and contrast SDTMs and ADaMs, and 3) main SDTM and ADaM components/summary. E-mail us for the latest course fee and registration. You may want to check your organization for class fee reimbursement.
Students start the self-study course based on their schedule and have access to real world SDTMs and ADaMs as well as SAS macros to create them. A student journal is also included to track your progress and any questions you may have. Get a CDISC certificate from Gupta Programming after completing the class.
Local to the Los Angeles or San Diego area? Contact me for an on-site customized SAS® class for any industry.
Contact me for client specific training on any topic below:
Practical R for SAS Programmers (New)
I. Most Popular Classes
Getting Started with Clinical SAS Programming
Data Science using SAS (New Three-Day Content, Instructor based Online class with limited students)
Standards for CDISC Compliance and Clinical Data Quality Checks Using SAS (New Hands-on class)
CDISC Mapping and Strategies - Three-Day content [Also for Medical Devices and Oncology Studies]
Sharpening Your SAS Skills: Preparing for Base Certification Run SAS Programs to confirm your SAS Skills
Sharpening Your Advanced SAS Skills: Preparing for Advanced Certification Become a SAS Guru
II. Getting started with SAS Classes
SAS® Essentials (Introduction to SAS® Programming)
Getting Started with SAS Enterprise Guide
Utilizing SAS Enterprise Guide for more effective SAS® Programming and Project Management
III. Advanced SAS Topic Classes
A to Z Analysis and Validation using Proc Tabulate
Automating Tasks using SAS Macro and Metadata Programming
Customized Reports with SAS Output Delivery System
Maximizing Productivity and Efficiency using Proc SQL
Quick Results with SAS Output Delivery System
Proc Report, Proc Template and ODS Unleashed
Publication Quality Tables using Proc Report
See Critic's Choice link for quotes from satisfied students

SAS Books (Top)
Output Delivery System or ODS is not only very powerful, but a must-know skill for all SAS®programmers mainly because it is so easy to apply and can have a big impact. Preparing for the SAS® Base Certification exam? Then review Sharpening Your SAS® Skills book.
Visit SAS's Bookstore to download a free book chapter and all the SAS® examples. Remember to e-mail us witih any questions you may have on these SAS®topics.
Quick Results with ODS Book ReviewSharpening Your SAS Skills Review Download/Purchase e-book

Download Popular SAS® e-Guides (Top)
Each e-Guide is a PDF file that is iPad compatible. SAS® Savvy Premium members get access to these SAS® e-Guides with an introduction video session for each. See SAS Savvy home page for a complete list of topic dates.
Not a SAS® Savvy Premium member? Download each SAS® e-Guide now for only $24.95 each. CDISC 101 Mapping e-guide is only $65. Requires Acrobat Reader 10.1.0 or greater for internal and external links to work. See SAS e-Guide bookstore.
Automating Tasks Using SAS® Macro Programming (Class Outline)
(40 pages with over 85 examples, internal and external hyperlinks)

"Well written. Great book for starters in SAS Programming.", Sandeep Kottam, Senior Statistical/CDISC SDTM Programmer
"Another excellent book which would be an excellent asset to the Macro programmers. Very detailed instructions in many areas which most SAS programmers, even experienced ones would not have been exposed to. Excellent instructions about the various uses of the different functionalities of SAS Macros. I would recommend this to even advanced programmers who want some simple and fast reference material instead of F1.", Murphy Choy, Risk Analyst, Standard Chartered Bank, Singapore
(Hardcopy available on LuLu for $24.95)

Compare and Conquer SAS® Programming Techniques (Top)
(22 pages of 25 tables, internal and external hyperlinks)

(Hardcopy available on LuLu for $24.95)

Quick Results with Proc SQL (Free Preview) (Class Outline) (Top)
(39 pages with over 75 examples, internal and external hyperlinks)
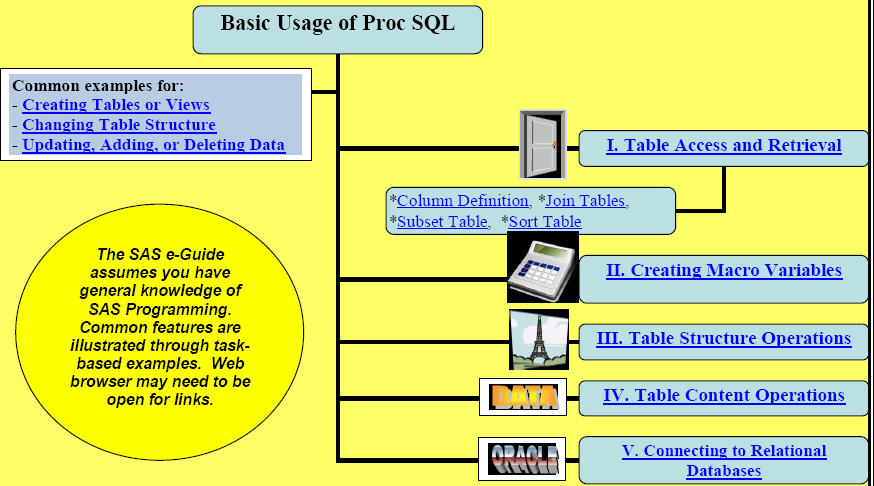
(Hardcopy available on LuLu for $24.95)

Quick Results with Proc Report (Class Outline) (Top)
(16 pages with over 10 examples, internal and external hyperlinks)
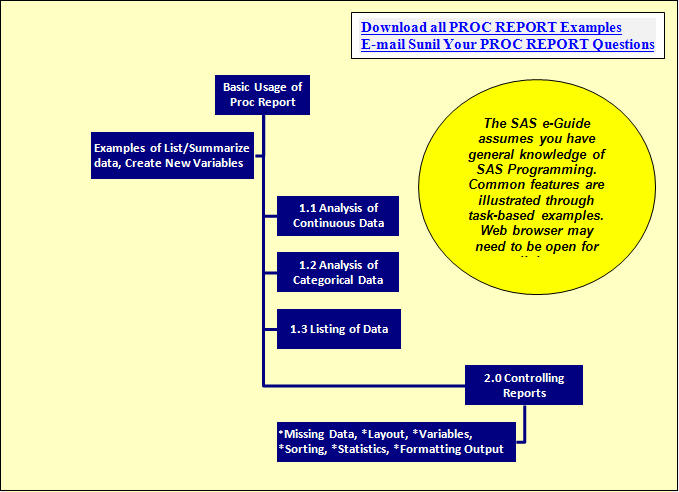
(Hardcopy available on LuLu for $24.95)

A to Z Analysis and Validation using Proc Tabulate (Class Outline) (Top)
(30 pages with over 25 examples, internal and external hyperlinks)
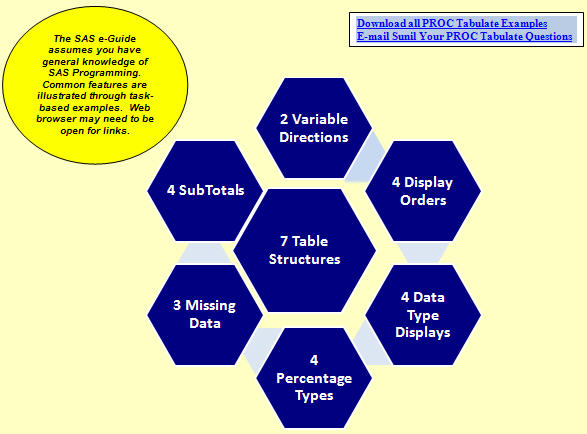
(Hardcopy available on LuLu for $24.95)

Pharmaceutical Industry On-Demand Classes (Top)
Need immediate access to a wealth of comprehensive information on understanding the uniqueness of the pharmaceutical industry and how SAS® statistical programmers play a vital role in assuring the quality of clinical data and statistical analysis? Select one or more of my specialized on-demand classes below and download the full class notes and SAS® examples.
View the two-day intense courses outline. View the complete five-webinar package.
Contact me for client specific training on any topic below:
1. Best Practices in Being 'On-Call' for FDA Submission Audits
2. Best Practices in SAS Statistical Programming for Regulatory Submission: Understanding and Applying the QC Plan to Validate Summary Tables
3. Best Practices in SAS Statistical Programming for Regulatory Submission: Creating Publication-Quality Summary Tables
4. Compare and Conquer SAS Programming Techniques for FDA Submissions
5. Effective Clinical Data Acceptance Testing
These courses are offered through The Center for Professional Advancement, an organization which is CEU accredited.

© 2011 Sunil Gupta, All Rights Reserved


